Ionomer Thin Films
Ionomer Thin Films
In many of the electrochemical energy conversion devices, an ionomer’s role is not limited to polymer-electrolyte membrane (PEM), and extends to the porous electrode structures. In such a heterogeneous structure, ionomers exist as nanometer-thick electrolyte “thin films” binding the catalytic particles and facilitating transport of ionic and gaseous species. Key is to develop model systems for thin films that can mimic electrode ionomer behavior, and help elucidate the interplay between nano-confinement and interactions at dynamic interfaces. Only then can one provide insight into tuning and optimization of ionomer film functionalities for energy conversion devices.
Ionomers: Bulk to Thin-Films
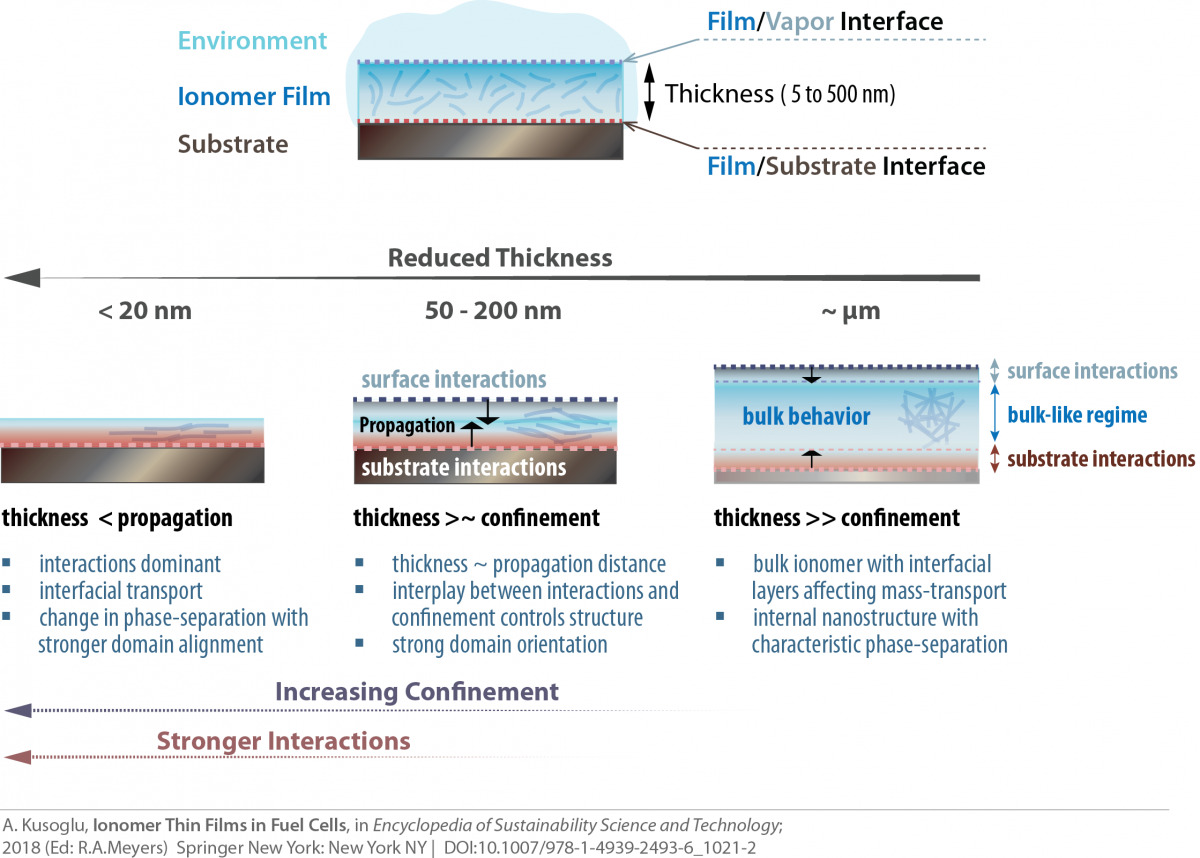
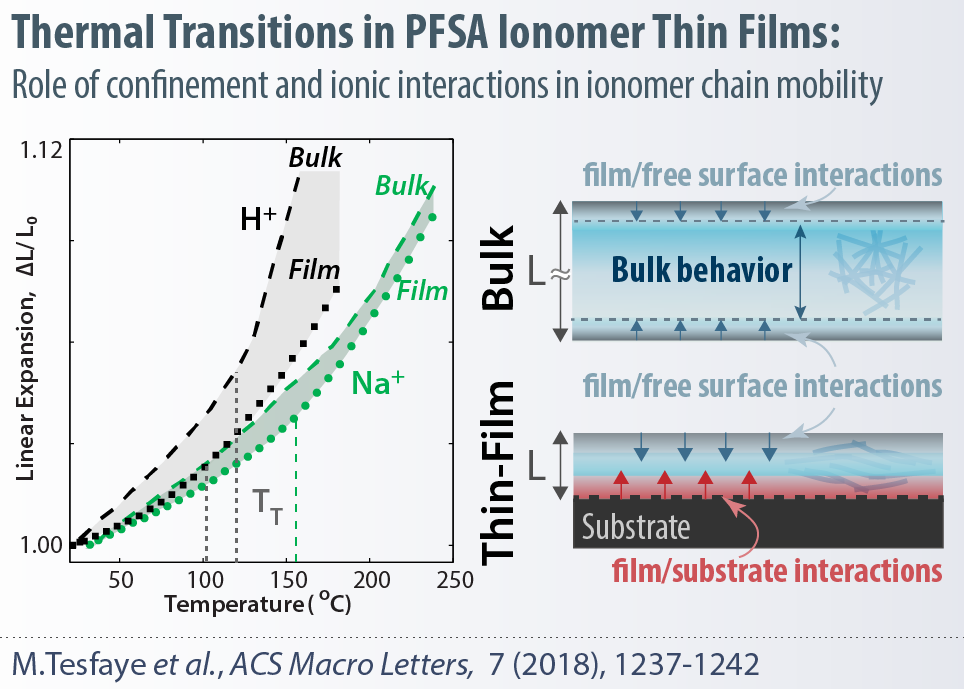
Ion-conductive polymers (ionomers) have been widely used as the solid-electrolyte in energy conversion devices, such as polymer-electrolyte fuel cells (PEFCs), alkaline fuel cells, redox flow batteries and electrolyzers, to facilitate selective transport of ionic and gaseous species. For advancing the efficiency and performance of these electrochemical systems, ionomers with improved stability and modified functionalities are needed both for membranes and catalyst layers. In fuel cells, for example, ionomers function as nanometer-thick electrolyte thin film to facilitate the transport of ionic and gaseous species to the catalytic particles. Once confined to these thicknesses (<100 nm) in a heterogeneous electrode structure, ionomer properties change drastically from the bulk (membrane) and are significantly more influenced by ionic and interfacial interactions.
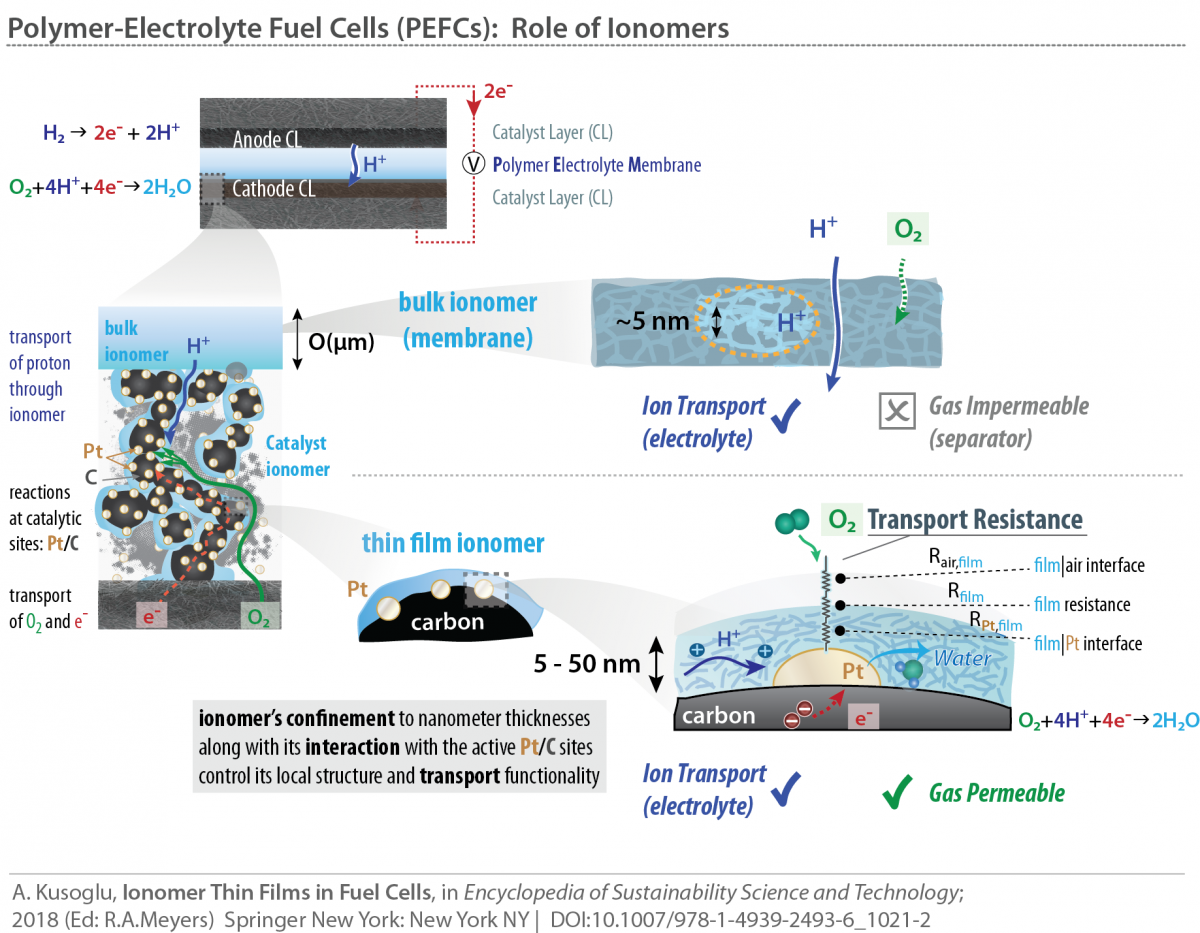
Throughout this thickness regime, ionomer chemistry, in particular, its pendant-chain structure and ion-exchange capacity, become even more influential on these interactions, which govern ionomer’s functionality and ultimately affects their performance in devices. Our group explores the structure-property relationships of several ionomers, including, but not limited to, perfluorosulfonic-acid (PFSA) in both bulk (membrane) and thin-film (electrode) regime to examine how chemistry and confinement control material interactions. In particular, the latter phenomenon of confinement has important implications for fuel-cell catalyst-layer performance, which could be elucidated and improved through the interplay between ionomer chemistry and interfacial interactions.
Recent and Current Research on Ionomer Thin Films
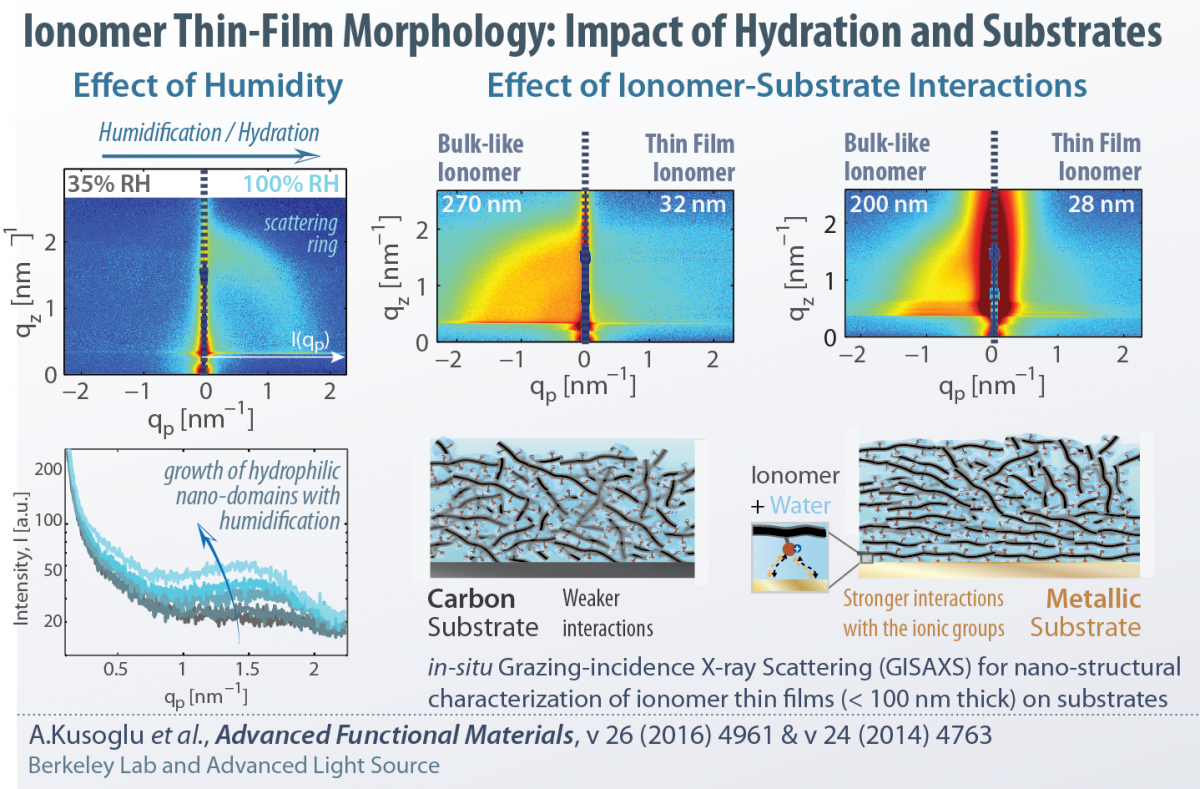
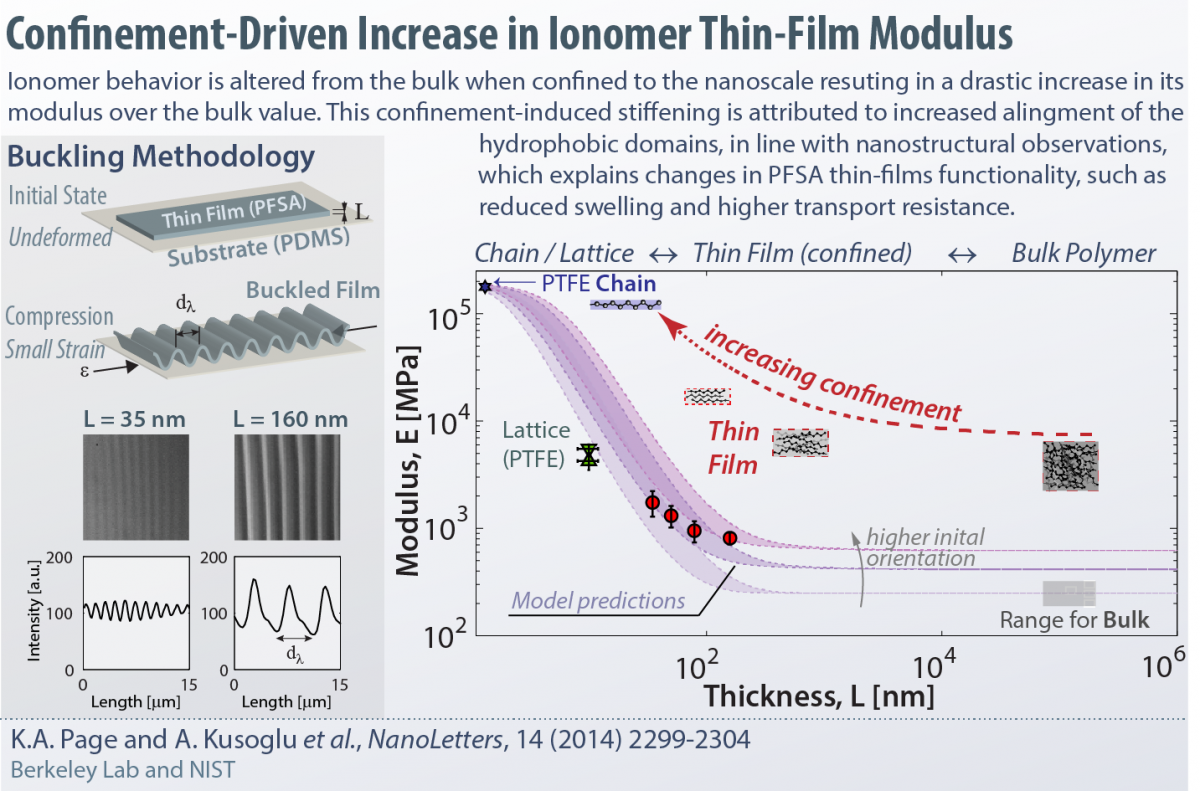
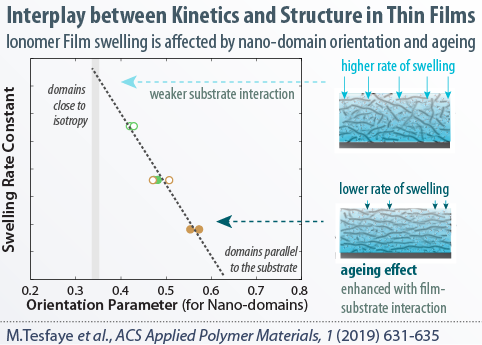
Ionomers, once confined to nanometer thicknesses, exhibit drastically different behavior compared to bulk. Our current research focus is to understand and optimize structure-functionality of thin-film ionomers, including, but not limited to, perfluorosulfonic acids (PFSAs). We develop various model systems to examine the ionomer thin-films under controlled environment, probe their morphology using advanced synchotron X-ray techniques (at the ALS), and employ advanced diagnostics techniques to measure their swelling, transport and mechanical properties on various substrate compositions. Some of our recent works related to this topic are:
-
Impact of substrate (Au, Pt, C, Si), thickness and processing effects on nanostructure and hydration behavior of PFSA thin films
-
Mechanical Characterization of Ionomer Thin Films under Confinement
-
Exploring the linkage between nanostructure and transport in PFSA thin-films
-
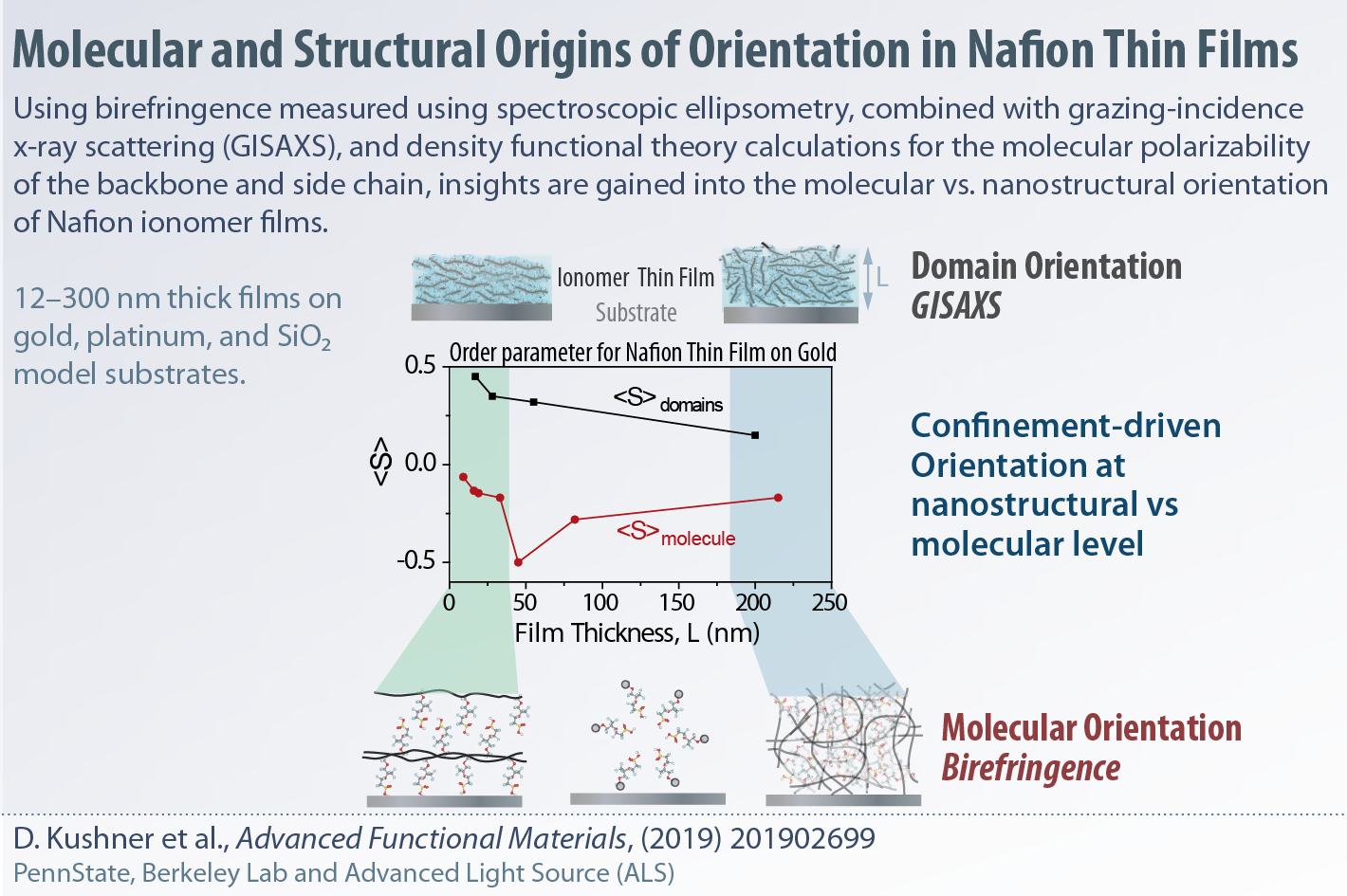
Elucidating the origins of structural and molecular orientation in thin films
-
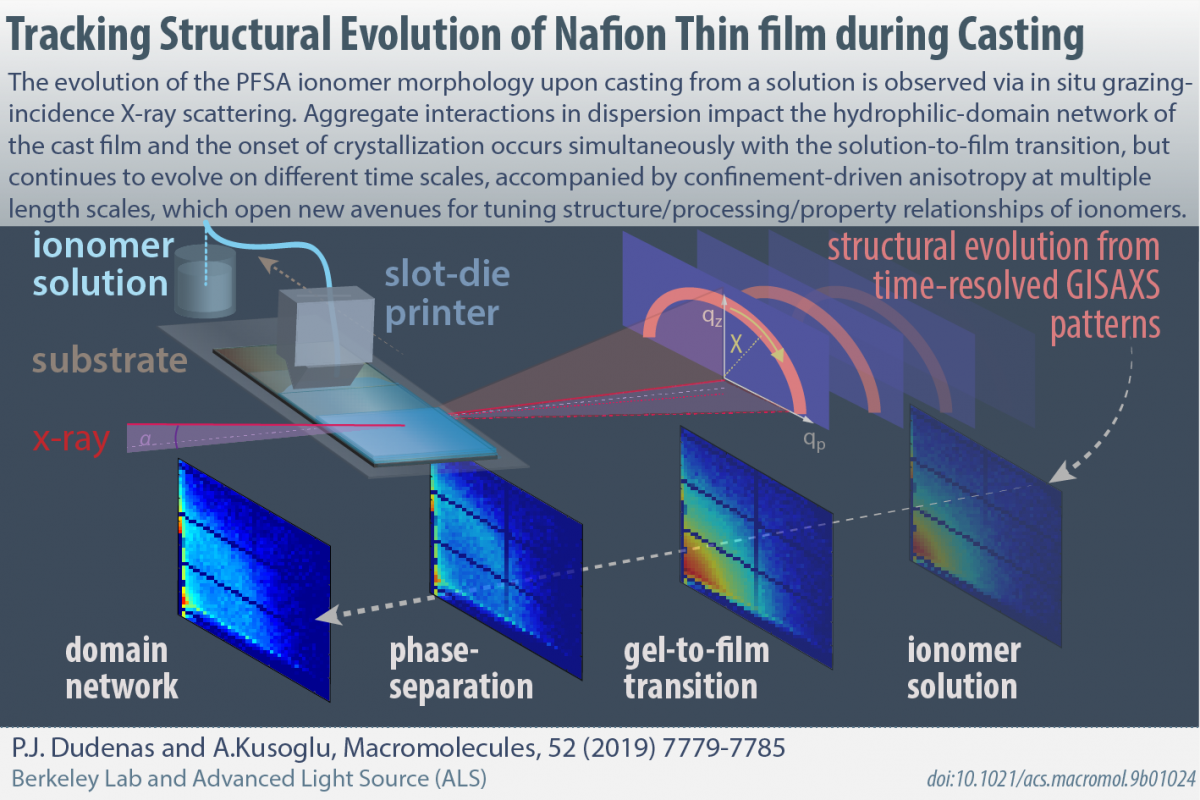
Monitoring ionomer film formation upon casting from a solution using novel in-situ x-ray techniques
-
Development of in-situ techniques for structure-property characterization of model thin-film systems
-
Establishing casting and processing protocols for fabricating thin-films of various ionomer chemistries
-
Investigation of chemistry and ion effects on thin-film behavior and properties