News
A new Compressive Creep Procedure for testing Polymer Membranes in Water
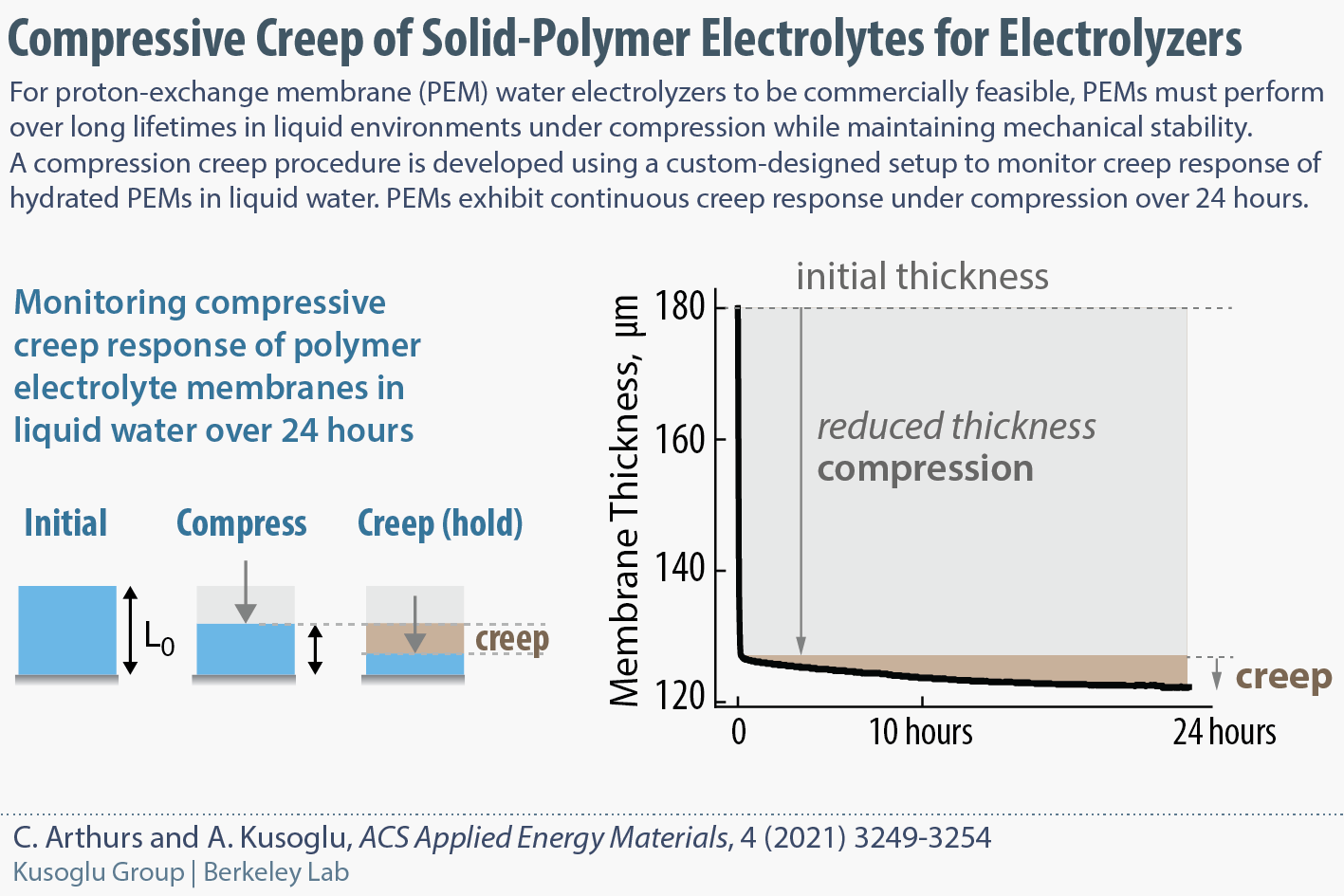 In water-splitting electrolyzers, the polymer electrolyte membrane is expected to exhibit stability under compressive loads for long operational times. To monitor the compressive-creep response of a membrane in liquid water, we have designed a new setup and developed a testing procedure.
In water-splitting electrolyzers, the polymer electrolyte membrane is expected to exhibit stability under compressive loads for long operational times. To monitor the compressive-creep response of a membrane in liquid water, we have designed a new setup and developed a testing procedure.
Our results show that PEMs exhibit continuous creep response under compression over 24 h, with a dependence on the applied pressure and hydration state. This work was performed under the HydroGEN and H2NEW consortia funded by DOE.
Anion-Exchange Ionomer Thin Films for Electrodes of Alkaline Devices
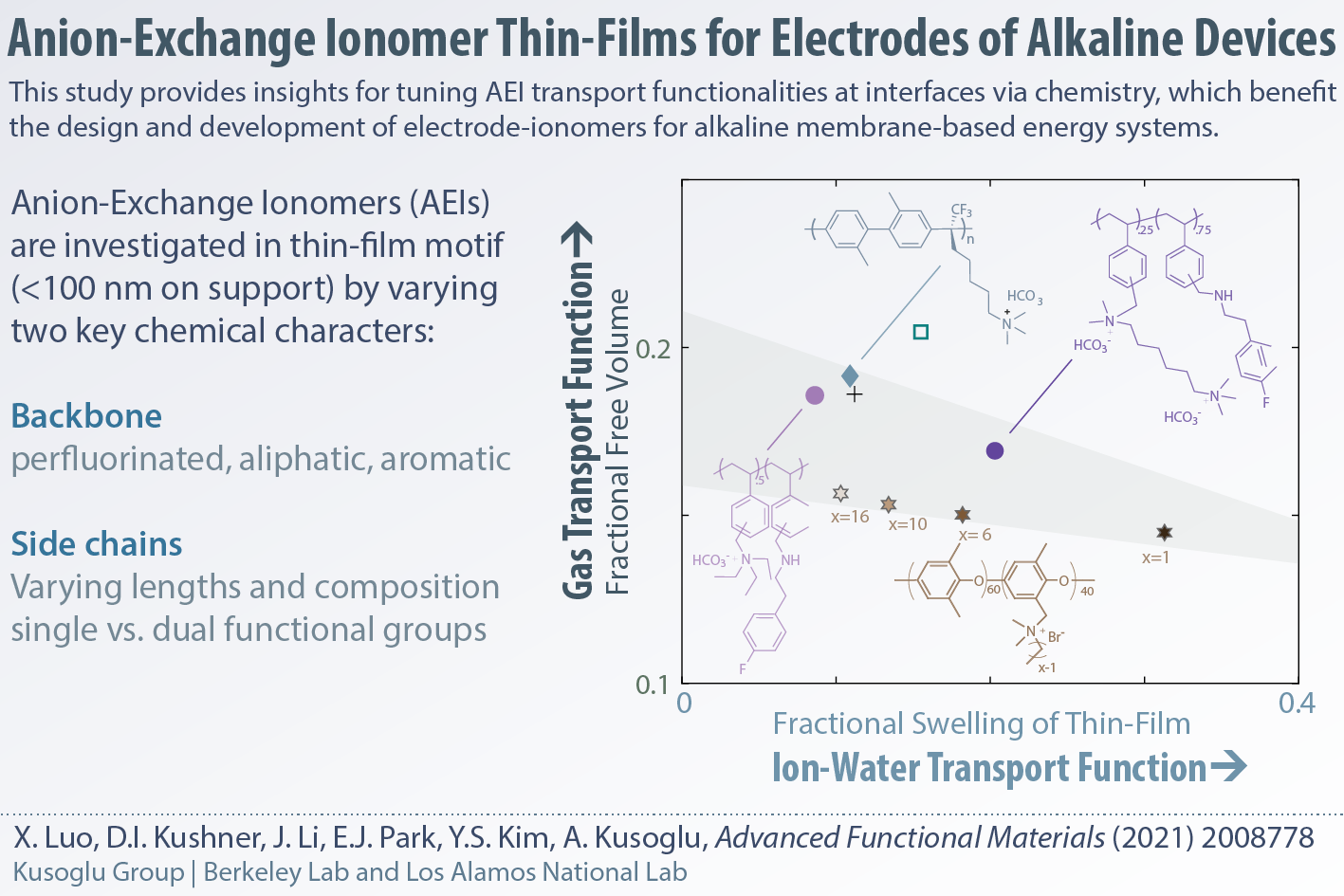 In collaboration with Yu Seung Kim's group in Los Alamos National Lab, we have investigated how anion-exchange ionomers (AEIs) change their behavior when cast as thin-films on supports compared to the bulk membranes. We have examined AEIs with different backbone chemistry as well as side-chain compositions to develop a better picture for the chemistry-anion effect on ionomer film properties for understanding their function in electrodes of alkaline devices. This work was carried out under the HydroGEN consortium of DOE.
In collaboration with Yu Seung Kim's group in Los Alamos National Lab, we have investigated how anion-exchange ionomers (AEIs) change their behavior when cast as thin-films on supports compared to the bulk membranes. We have examined AEIs with different backbone chemistry as well as side-chain compositions to develop a better picture for the chemistry-anion effect on ionomer film properties for understanding their function in electrodes of alkaline devices. This work was carried out under the HydroGEN consortium of DOE.
Effect of Cerium Ions on Structure-Property Relationship of Ionomer Membranes
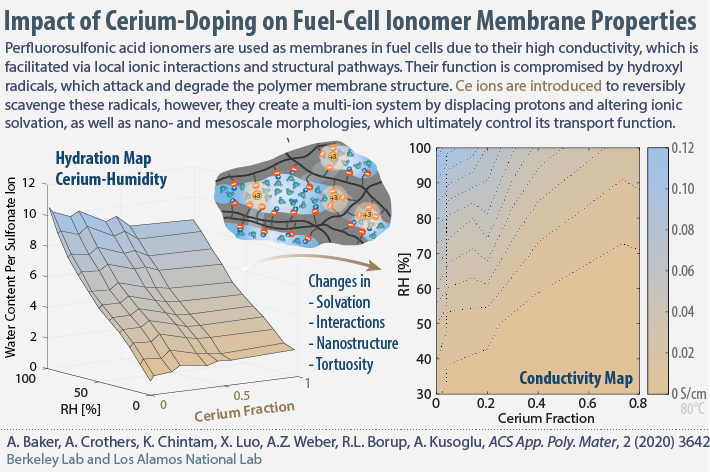
In collaboration with Los Alamos National Lab, we have published a paper that systematically investigates and explains how the incorporation of Cerium (III) and Cerium(IV) cations into a proton-exchange ionomer membrane alters the internal structure, solvation, and hydration properties, as well as ion conductivity. The combined experimental and theoretical analysis reveals a nonlinear change in conductivity driven by cation solvation at the molecular level and morphological changes at longer length scales. The results are used to construct various property-maps for the ionomer membrane.
Transport phenomena in flow battery ion-conducting membranes
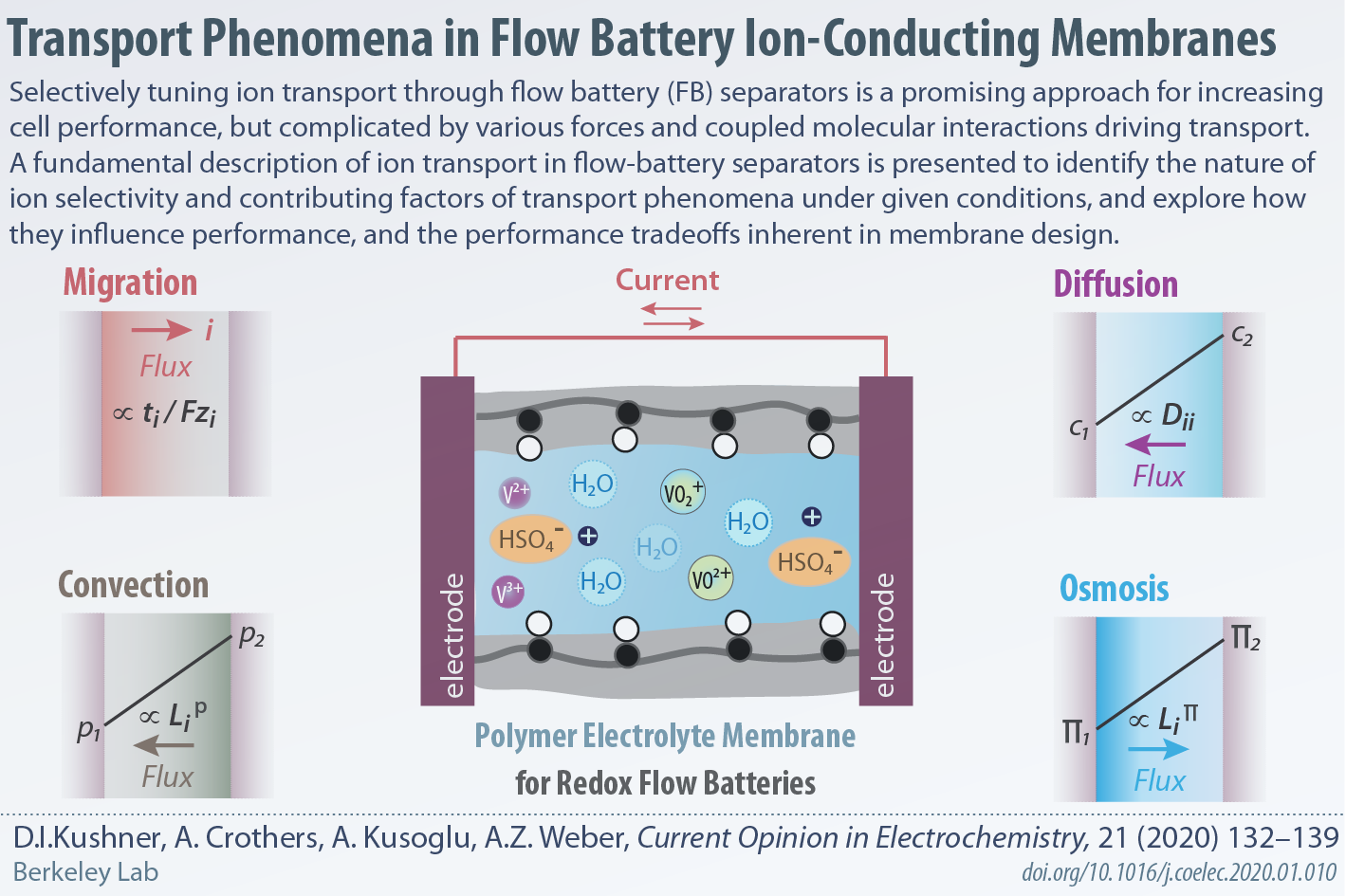 In collaboration with Weber group, we have published an Opinion Article in Current Opinion in Electrochemistry that discusses various contributing factors of transport phenomena, how they influence cell performance, and the performance tradeoffs inherent in membrane design.
In collaboration with Weber group, we have published an Opinion Article in Current Opinion in Electrochemistry that discusses various contributing factors of transport phenomena, how they influence cell performance, and the performance tradeoffs inherent in membrane design.
ALS Highlight: Multimodal Study of Ion-Conducting Membranes
Our work on multi-modal characterization of ionomers has been selected as a science highlight by the Advanced Light Source. This study uses multiple x-ray characterization tools to elucidate the chemical and structural origins of the improved performance of novel ionomers from 3M Company. The work provides insight into factors impacting the proton conductivity of ionomers used as solid-electrolyte membrane in fuel cells and water-splitting devices.
Journal Publication: Characterization of Anion-Exchange Membranes
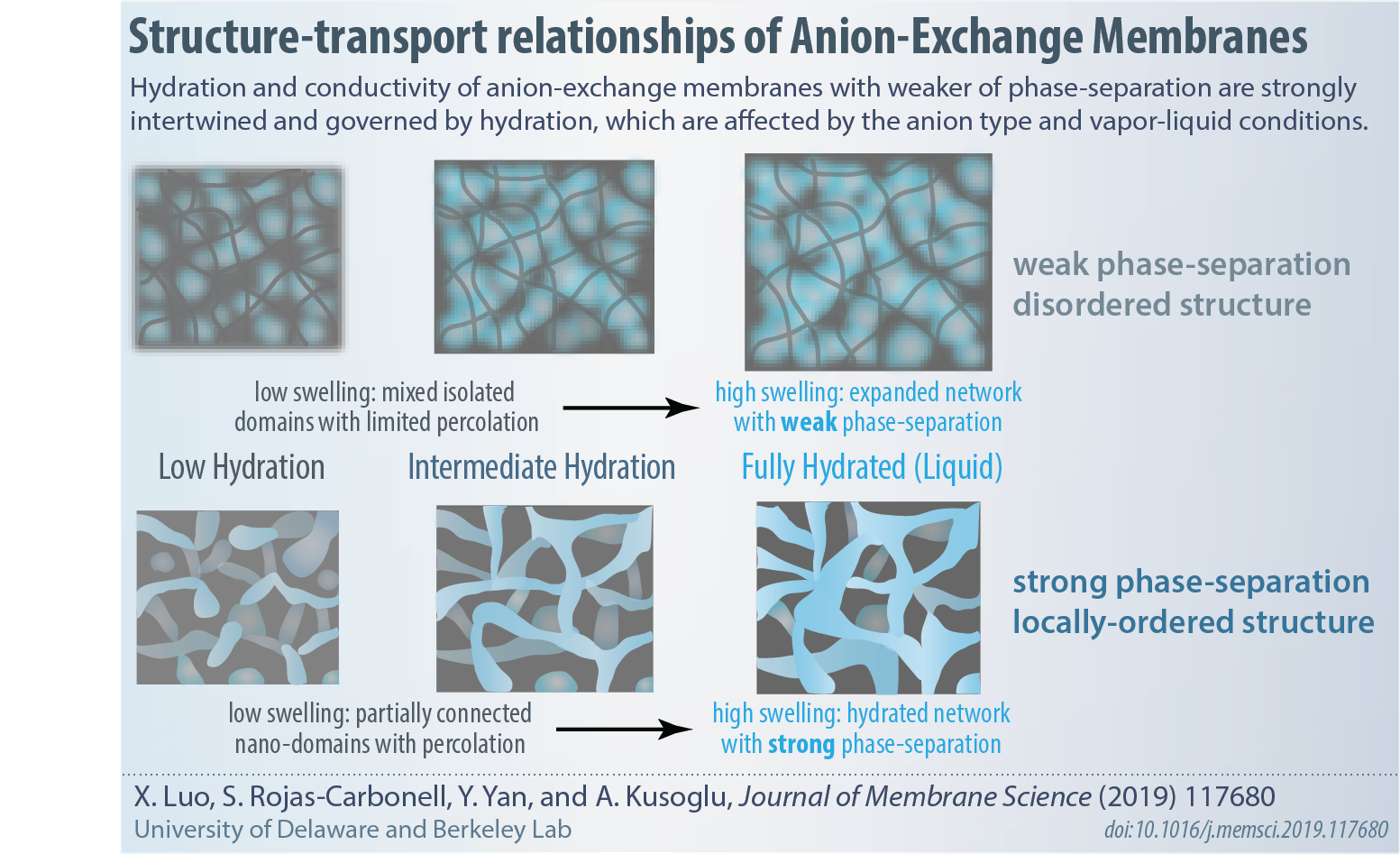 A systematic study on the effects of anions and hydration on properties of AEMs based on commerically available polymers as well as a PAP-based polymer, provided by Yushan Yan Group at University of Delaware.
A systematic study on the effects of anions and hydration on properties of AEMs based on commerically available polymers as well as a PAP-based polymer, provided by Yushan Yan Group at University of Delaware.
Review Paper: New Insights into PFSA Ionomers
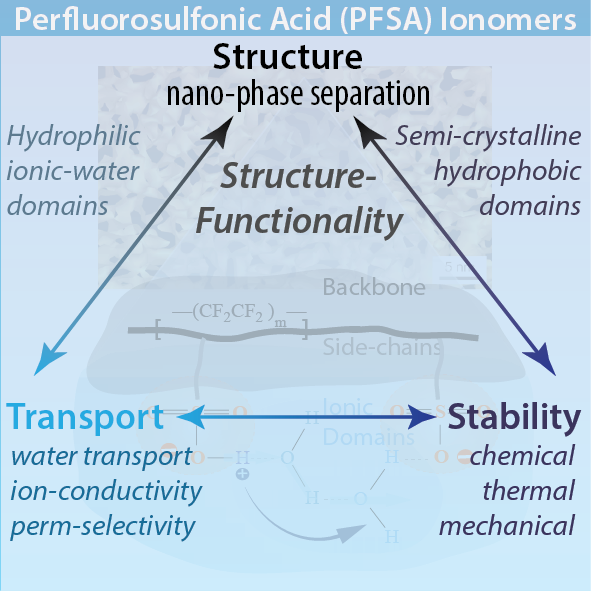 In this comprehensive review, recent progress and developments on perfluorinated sulfonic-acid (PFSA) membranes have been summarized on many key topics. Although quite well investigated for decades, PFSA ionomers’ complex behavior, along with their key role in many emerging technologies, have presented significant scientific challenges but also helped create a unique cross-disciplinary research field to overcome such challenges. Research and progress on PFSAs, especially when considered with their applications, are at the forefront of bridging electrochemistry and polymer (physics), which have also opened up development of state-of-the-art in situ characterization techniques as well as multiphysics computation models.
In this comprehensive review, recent progress and developments on perfluorinated sulfonic-acid (PFSA) membranes have been summarized on many key topics. Although quite well investigated for decades, PFSA ionomers’ complex behavior, along with their key role in many emerging technologies, have presented significant scientific challenges but also helped create a unique cross-disciplinary research field to overcome such challenges. Research and progress on PFSAs, especially when considered with their applications, are at the forefront of bridging electrochemistry and polymer (physics), which have also opened up development of state-of-the-art in situ characterization techniques as well as multiphysics computation models.
This review covers and summarizes ~1000 papers on PFSAs in the open literature.
This paper is published under open-access and can be downloaded from the journal webpage.
Journal Publication: Probing Morphology of Sulfonated Ionomers via Tender X-rays
 In this paper, our group and researchers at the Advanced Light Source (ALS) collaborated to examine the morphological features of sulfonated ionomers, including novel multi-acid side-chain chemistries developed by 3M, using state-of-the-art in-situ x-ray scattering and spectroscopy technqiues with tunable energy (eV) range, such as Sulfur-edge.
In this paper, our group and researchers at the Advanced Light Source (ALS) collaborated to examine the morphological features of sulfonated ionomers, including novel multi-acid side-chain chemistries developed by 3M, using state-of-the-art in-situ x-ray scattering and spectroscopy technqiues with tunable energy (eV) range, such as Sulfur-edge.
Our work has been featured on the cover of JACS.
Journal Publication: Multi-acid Side-Chain Ionomers with High Proton Conductivity
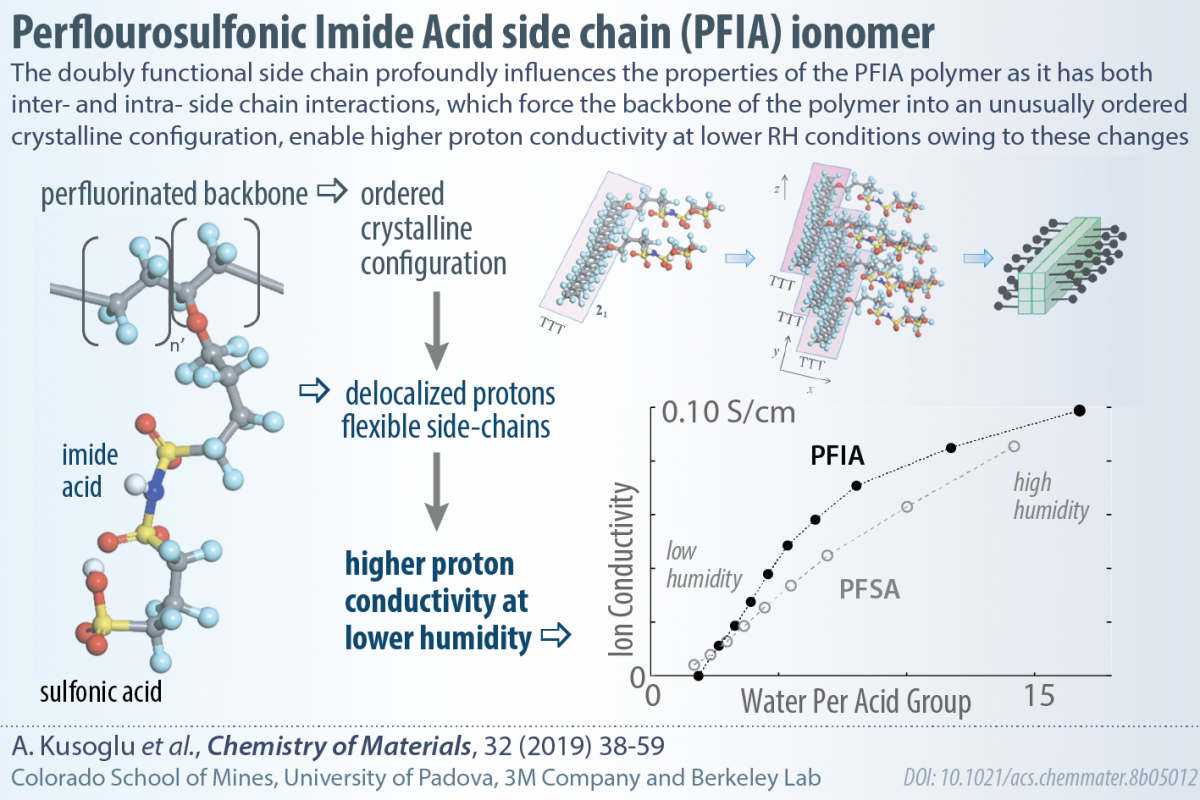 We have published a comprehensive paper exploring and elucidating the effect of complex side-chains on the properties of polymer-electrolyte membranes, in collaboration with Herring Group, Vito Di Noto, and 3M. This contribution is a result of a unique collaboration of researchers working on polymers, structural characterization, transport phenomena and theoretical modeling. Our results show how the side-chain modification of an ionomer can have unintended consequences on membrane properties.
We have published a comprehensive paper exploring and elucidating the effect of complex side-chains on the properties of polymer-electrolyte membranes, in collaboration with Herring Group, Vito Di Noto, and 3M. This contribution is a result of a unique collaboration of researchers working on polymers, structural characterization, transport phenomena and theoretical modeling. Our results show how the side-chain modification of an ionomer can have unintended consequences on membrane properties.
Journal Publication: Characterization of PFSA Ionomer with Elastin-like Polypeptide
Tuning the properties of PFSA ionomer thin films on catalystic surfaces such as Pt is an important step toward controlling their functionality in electrodes of fuel cells and electrolyers. Renner Group (CaseWestern) works on altering the Ionomer-Pt interface by modulating the morphology of Nafion ionomer by incorporating an elastin-like polypeptide (ELP). We have collaborated to accomplish the structural characterization of these ionomers using x-ray techniques at the ALS, which have helped elucidate the origins of the observed functionality changes at the ionomer interface.
Journal Publication: Molecular and Nanostructural Orientation of Nafion Thin Films
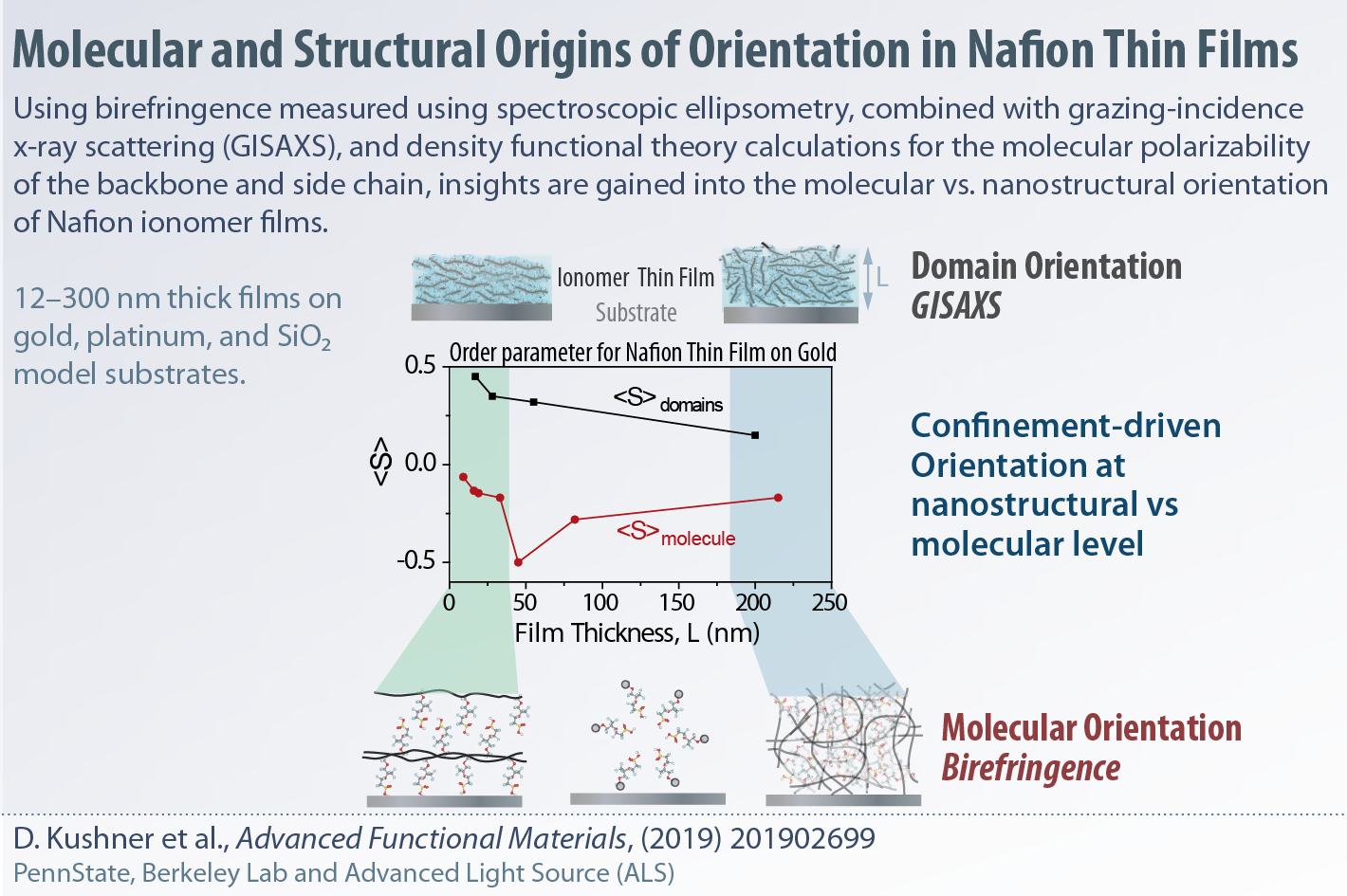 In collaboration with Hickner Group (PennState), we have investigated the confinement-driven orientation in Nafion thin films to delineate the molecular orientation and nanostructural orientation (from GISAXS). The degree of orientation at these lengthscales differ as the ionomer gets thinner and depends on the nature of the substrate composition.
In collaboration with Hickner Group (PennState), we have investigated the confinement-driven orientation in Nafion thin films to delineate the molecular orientation and nanostructural orientation (from GISAXS). The degree of orientation at these lengthscales differ as the ionomer gets thinner and depends on the nature of the substrate composition.
Book Chapter: Ionomer Thin Films in Fuel Cells
 This chapter on the role of ionomers as thin films in the electrodes of fuel cells is published in Encyclopedia of Sustainability Science and Technology.
This chapter on the role of ionomers as thin films in the electrodes of fuel cells is published in Encyclopedia of Sustainability Science and Technology.
In polymer-electrolyte fuel cells (PEFCs), ionomers play a key role not only as a proton exchange membrane (PEM) but also as nanometer-thick electrolyte “thin films” within porous catalyst layer (CL) structures, where they bind and cover the catalytic particles and provide transport pathways for the ions and reactant species. As the ionomer is confined to nanometer thicknesses in the CLs, its intrinsic nano-morphology and resulting transport properties deviate from bulk membrane behavior. In thin-film form, the ionomer forms dynamic interfaces with the air and substrate, which impose stronger impact on ionomer’s structure/functionality. This interplay between the confinement and dynamic interactions controls the catalyst ionomer’s properties, which affects the local transport resistances in CLs, and ultimately PEFC performance.
Journal Publication: Simulating crack growth in fuel-cell membranes
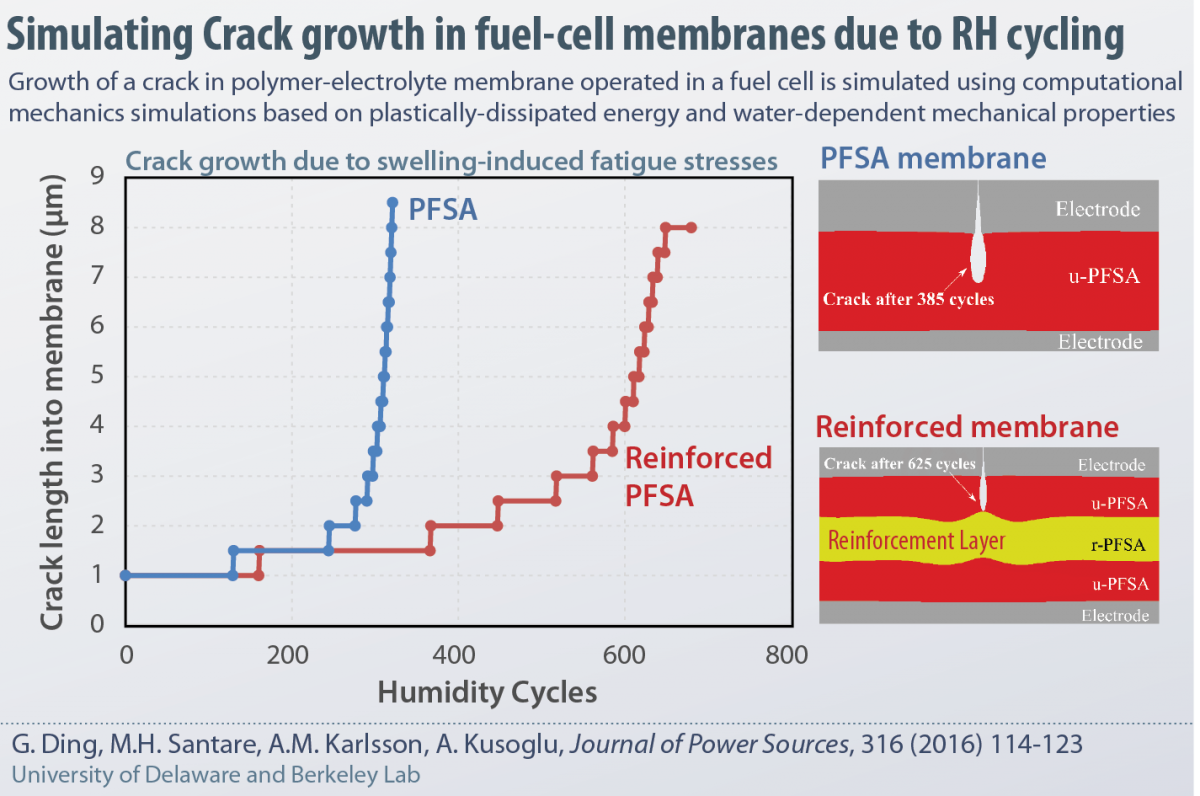 The role of cyclic hygrothermal stressors in generating an alternating stress-state in fuel-cell membranes has been inferred in many studies. Yet, no study has demonstrated how an existing crack in a fuel cell membrane grows under cyclic loads. In this study, in collaboration with researchers from University of Delaware, a computation mechanics model of a fuel cells is developed based on the theory of plastically-dissipated energy during crack growth, which is simulated using finite element model. The model is capable of not only capturing the growth of an exsiting crack as a function of humidity cycles, but also demonstrating the role of mechanical reinforcement in slowing down the crack growth and extending membrane lifetime, in line with experimental observations.
The role of cyclic hygrothermal stressors in generating an alternating stress-state in fuel-cell membranes has been inferred in many studies. Yet, no study has demonstrated how an existing crack in a fuel cell membrane grows under cyclic loads. In this study, in collaboration with researchers from University of Delaware, a computation mechanics model of a fuel cells is developed based on the theory of plastically-dissipated energy during crack growth, which is simulated using finite element model. The model is capable of not only capturing the growth of an exsiting crack as a function of humidity cycles, but also demonstrating the role of mechanical reinforcement in slowing down the crack growth and extending membrane lifetime, in line with experimental observations.
Confinement-Driven Increase in Ionomer Thin-Film Modulus
Our group, in collaborati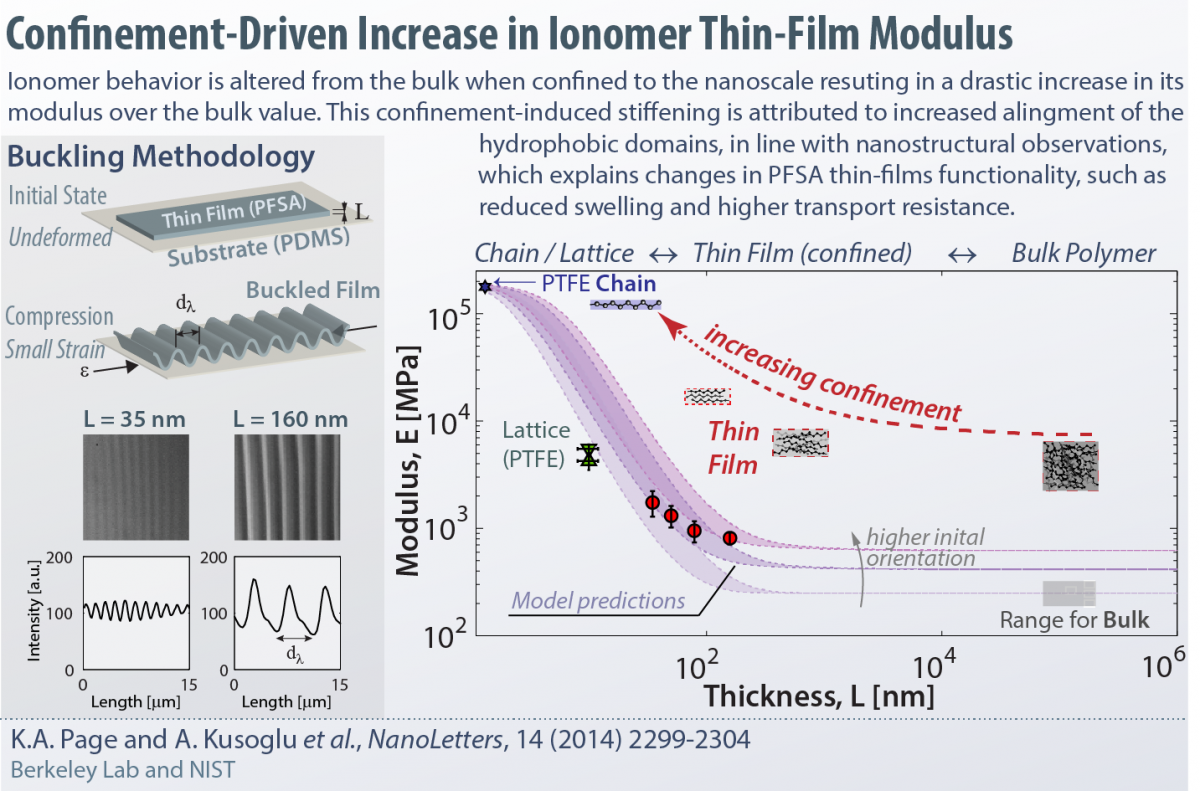 on with NIST, demonstrated the applicability of the buckling methodology to the ionomer thin films for measuring their moduli. In this work, we present a compelling evidence for the confinement-driven increase in stiffness of Nafion thin film due to nanostructural changes. As the ionomer is confined onto nanometer thicknesses, the increased orientation of its nano-domains results in a higher resistance to deformation in the plane of the film, thereby increasing its modulus compared to bulk values.
on with NIST, demonstrated the applicability of the buckling methodology to the ionomer thin films for measuring their moduli. In this work, we present a compelling evidence for the confinement-driven increase in stiffness of Nafion thin film due to nanostructural changes. As the ionomer is confined onto nanometer thicknesses, the increased orientation of its nano-domains results in a higher resistance to deformation in the plane of the film, thereby increasing its modulus compared to bulk values.